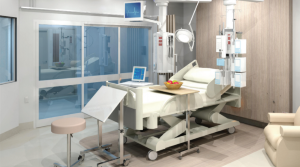
Types A, B and C for Advanced Healthcare Systems.

Download: Type A 32 Bed Modular Plan

Download: Type B 52 Bed Modular Plan

Download: Type C 64 Bed Modular Plan
Type D and E for Developing Healthcare Systems.

Download: Type D 64 Bed Modular Plan

Download: Type E 92 Bed Modular Plan
HPI is a recognised International specialist in Health Facility Planning, Design and Modular Prefabrication. HPI works throughout the world with a focus on developing Countries. HPI is a prolific author of open sourced Standards and Guidelines for Health Facility Design such as the popular International Health Facility Guidelines (iHFG).
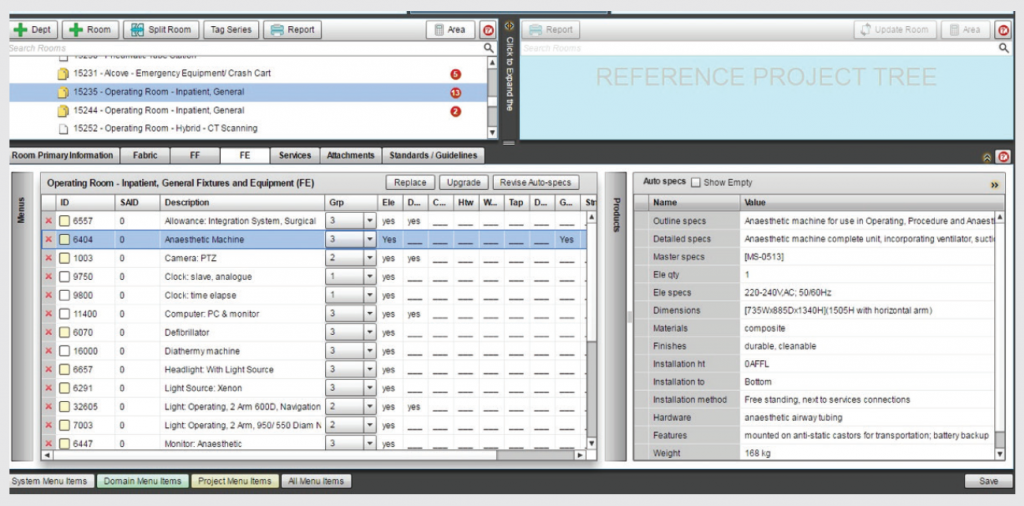
HPI has customised iHFG for use by 8 Health Authorities around the world. HPI is also the distributor and customizer of a range of professional web-based Health Planning software known as the HFBS platform.
HPI has activities in 34 Countries and projects in 20. HPI has been responsible for over 300 healthcare projects world-wide. HPI staff and management feel obliged to make a contribution to the world in the current Covid-19 Pandemic. Therefore we contribute the most valuable thing we have, a gift of knowledge.
HPI has produced a series of open-sourced Quarantine Hospital Modular Designs in response to the Coronavirus (COVID-19) epidemic. The 5 design models comply with the principles of Infection Control and Isolation as per iHFG – International Health Facility Guidelines Part D. The different models should suit the different circumstances of different Countries.
These facilities are designed for conventional or modular prefabrication by whatever means available in different Countries.
If the capabilities do not exist, please contact HPI and we may be able to organize it in your Country.
The Goals of HPI’s Quarantine Hospital Modular Design models are:
Short Term Phase 1
Quickly erect health facilities that can hold large numbers of patients in quarantine conditions. In the current pandemic, extraordinary measures have been taken and are being taken to assure the health authorities ability to provide the care needed where and when it is needed. From the large-scale preparations jointly with the defense departments, to the numerous smaller scale initiatives, at times unavoidably disruptive, modifications to existing facilities to augment the capacities of the local ICUs. As a specialist in Health Planning and author of numerous infection control and isolation guidelines we, at HPI believe what is required is a series of rapidly built Quarantine hospitals with proper negative pressure isolation facilities complying with iHFG. This is the short term aim of the 5 modular quarantine hospital designs provided. These facilities would supplement existing hospitals. If these are build using modular prefabrication techniques, they can be placed in carpark areas close to existing hospitals then after the pandemic they can be relocated one module at a time to a new, permanent location to act as permanent Community Hospitals but ready for the next pandemic.
Short Term Benefits
- Proper isolation of patients as per recognised standards to fight the Covid-19 Pandemic
- Reasonable protection for medical, nursing and support staff
- No compromise on healthcare standards one would expect in a regular hospital
- Departure from the converted exhibition halls or assembly halls turned into field hospitals with all the expected risks and compromises
Addressing these issues using HPI’s Quarantine Hospital Design Framework will allow moving to the medium term needs in a faster and more structured way.
Long Term Phase 2
Looking ahead to the end of 2020 and the expected control of Covid-19 around the world, these facilities can be relocated to other locations within a couple of months and installed permanently as community hospitals with 64+ beds each.
Additional support services can be provided to make them fully self-sufficient.
Seen this way the investment will not be wasted but re-purposed for the long term general health of the population.
Nothing in the design or construction of these facilities is either temporary of sub-standard. So, the long term benefit is assured.
Long Term Benefits
Use emergency Covid-19 Pandemic Government investments for the long term improvements to healthcare
Have the facilities ready for the next pandemic as they are optimized for negative pressure respiratory isolation.
These facilities can return to their original use as Quarantine Hospitals within a few days.
HPI makes it easier to quickly move forward
The Challenge, in the midst of this current crisis, is to move forward rapidly, whilst at the same time researching what standards to follow and reviewing how planning happens across healthcare systems in the medium and long term.
HPI’s 5 Quarantine Hospital models aim to provide a ready-to-use and free solution.
For a description of the Isolation requirements refer to iHFG Part D
For the Engineering guidelines on Isolation facilities refer to iHFG Part E
If you have technical questions refer them to iHFG Techsupport
If you required professional assistance with these 5 designs or with your own initiatives, refer to HPI, the author of iHFG.